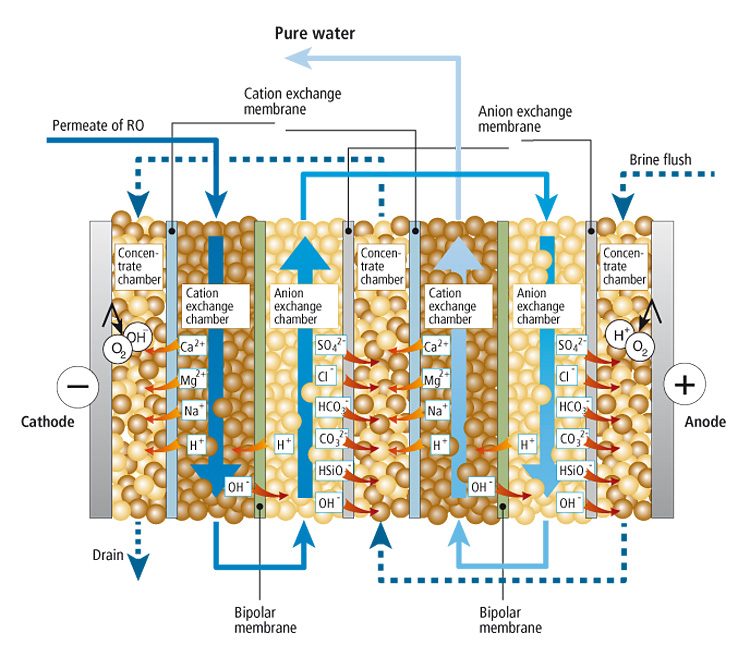
Technologie der Elektro-Deionisation
Over the past decades, the use of Electro-Deionization modules (EDI) has established itself as a proven technology for producing high-purity water. The desire to eliminate regeneration chemicals in post-treatment systems such as ion exchange units used in combination with reverse osmosis (RO) systems has significantly driven the adoption of this technology.
EDI cells have increasingly replaced conventional ion exchangers as polishers requiring external regeneration. With EDI, there is no need for regular replacement of DI cartridges, thereby avoiding fluctuations in product water quality.
Key Advantages of EDI Technology:
- Continuous Operation: EDI cells operate without interruptions for regeneration or DI cartridge replacement.
- Consistent Water Quality: Ensures stable, high-quality product water at all times.
- Sustainability: EDI requires no chemicals for regeneration, making it an environmentally friendly and cost-efficient solution.
- Cost Efficiency: Eliminating the need for chemicals and DI cartridge changes significantly reduces operating costs.
EDI cells remove ions from water that has typically been pre-treated by a reverse osmosis system or other purification technologies. EL-DI modules provide ultrapure water of the highest quality, up to 0.055 µS/cm (equivalent to 18.2 MΩ·cm) and can operate both continuously and intermittently.
Our EL-DI Technologies: MB Series and HD Series
EL-DI GmbH offers two specific technologies tailored to different application requirements:
EL-DI HD Series – For Demanding Applications
The innovative HD-Series is engineered for high-end requirements. It is based on mixed-bed technology and modern bipolar ion exchange membranes.
- High Tolerance: Operates even with elevated CO₂-levels of up to 30 ppm (depending on model type) and feedwater conductivity equivalent (FCE) of up to 80 µS/cm.
- Maximum Performance: The most compact and powerful cells on the market.
- Energy Efficient: Significantly lower energy consumption compared to other systems.
- Safe Operation: Operates at low voltage, eliminating the need for additional personal safety measures.
- Simplified Handling: No need for electrolytes, electrode rinsing, circulation pumps, and in many cases, no degassing or pH adjustment required.
EL-DI MB Series – Cost-Effective Solution for Standard Applications
The MB Series is a more economical alternative, specifically developed for standard applications under typical water conditions. It is based on mixed-bed chambers and monopolar ion exchange membranes.
- Cost Efficiency: High-quality modules at a particularly attractive price point.
- Reliable Performance: Ideal for common applications with consistent water quality..
- Safe Operation: Operates at low voltage, eliminating the need for additional personal safety measures.
- Simplified Handling: No need for electrode rinsing, circulation pumps, or— in many cases—pH adjustment.
Please click to enlarge.
Functional Diagram of the EL-DI Modules
We’re happy to answer your questions or take your orders via email at info@el-di.de or via phone at +49 40 35703877.